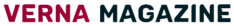The world’s greatest COVID-19 immunization study got in progress Monday with the first of 30,000 arranged volunteers assisting with testing shots made by the U.S. government – one of a few applicants in the last stretch of the worldwide antibody race.
There’s still no assurance that the test antibody, created by the National Institutes of Health and Moderna Inc., will truly ensure.
The required evidence: Volunteers won’t know whether they’re getting the genuine shot or a fake rendition. After two dosages, researchers will intently follow which gathering encounters more contaminations as they approach their day by day schedules, particularly in regions where the infection despite everything is spreading unchecked.
“Tragically for the United States of America, we have a lot of contaminations at this moment” to get that answer, NIH’s Dr. Anthony Fauci as of late told The Associated Press.
Moderna said the immunization was done in Savannah, Georgia, the primary site to get in progress among in excess of seven dozen preliminary destinations spread around the nation.
A few different immunizations made by China and by Britain’s Oxford University not long ago started littler last stage tests in Brazil and other hard-hit nations.
In any case, the U.S. requires its own trial of any immunization that may be utilized in the nation and has set a high bar: Every month through fall, the administration supported COVID-19 Prevention Network will reveal another investigation of a main competitor – every one with 30,000 recently selected volunteers.
The huge investigations aren’t simply to test if the shots work — they’re expected to check every possible antibody’s wellbeing. Also, keeping a similar report rules will let researchers in the end analyze all the shots.
Next up in August, the last investigation of the Oxford shot starts, trailed by plans to test an applicant from Johnson and Johnson in September and Novavax in October – if all goes as indicated by plan. Pfizer Inc. plans its own 30,000-man study this late spring.
That is a shocking number of individuals expected to focus in for science. However, lately, in excess of 150,000 Americans rounded out an online vault flagging interest, said Dr. Larry Corey, a virologist with the Fred Hutchinson Cancer Research Institute in Seattle, who regulates the examination locales.
“These preliminaries should be multigenerational, they should be multiethnic, they have to mirror the decent variety of the United States populace,” Corey told an antibody meeting a week ago. He focused on that it’s particularly imperative to guarantee enough Black and Hispanic members as those populaces are hard-hit by COVID-19.
It regularly takes a long time to make another immunization without any preparation, however researchers are setting speed records this time around, prodded by information that inoculation is the world’s best expectation against the pandemic. The coronavirus wasn’t known to exist before late December, and antibody creators got a move on. 10 when China shared the infection’s hereditary succession.
Only 65 days after the fact in March, the NIH-made antibody was tried in individuals. The principal beneficiary is urging others to chip in now.
“We as a whole vibe so vulnerable at the present time. There’s next to no that we can do to battle this infection. What’s more, having the option to take an interest in this preliminary has given me a feeling of, that I’m accomplishing something,” Jennifer Haller of Seattle told the AP. “Be set up for a great deal of inquiries from your loved ones about how it’s going, and a ton of thank-you’s.”
That first-stage study that included Haller and 44 others showed the shots fired up volunteers’ invulnerable frameworks in manners researchers expect will be defensive, with some minor reactions, for example, a concise fever, chills and torment at the infusion site. Early testing of other driving up-and-comers have had likewise reassuring outcomes.
On the off chance that everything goes directly with the last investigations, it despite everything will take a very long time for the principal information to stream in from the Moderna test, trailed by the Oxford one.
Governments around the globe are attempting to reserve a huge number of portions of those driving competitors so if and when controllers favor at least one antibodies, inoculations can start right away. However, the most readily accessible portions will be proportioned, probably held for individuals at most noteworthy hazard from the infection.
“We’re hopeful, mindfully idealistic” that the immunization will work and that “close to the furthest limit of the year” there will be information to demonstrate it, Dr. Stephen Hoge, leader of Massachusetts-based Moderna, told a House subcommittee a week ago.
Up to that point, Haller, the volunteer inoculated back in March, wears a cover openly and takes the equivalent separating precautionary measures prompted for everybody – while trusting that one of the shots in the pipeline works out.
“I don’t have the foggiest idea what the odds are this is the specific right antibody. However, thank heavens that there are such a significant number of others out there engaging this at the present time,” she said.