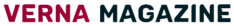On December 1, a man from Mount Vernon became the first person to receive lecanemab, a novel medication for Alzheimer’s disease, at the Memory and Brain Wellness Center at the University of Washington (UW) Medicine.
Professor of radiology and neurology at the University of Washington School of Medicine Dr. Thomas Grabowski remarked, “I’m pleased that Harborview Medical Center is one of the first West Coast hospitals to administer the IV infusion treatment to our qualifying patients.”
According to UW Medicine, Doug Davidson was the first patient at Harborview Medical Center to receive the “breakthrough” medication that has been demonstrated to slow cognitive decline in individuals with early Alzheimer’s disease.
The U.S. Food and Drug Administration authorized lecanemab in the summer of 2023. According to UW, it’s the first FDA-approved medication proven to alter the course of Alzheimer’s.
“This is still a step in the right direction, even though the outcome was relatively modest (a 27% slowing of cognitive decline and functional decline over 18 months),” stated Dr. Michael Rosenbloom, director of clinical trials at the Memory and Brain Wellness Center and associate professor of neurology at the UW School of Medicine.
According to UW, people with extremely mild Alzheimer’s dementia or mild cognitive impairment and amyloid plaques are eligible to utilize lecanemab.
Individuals who meet the eligibility requirements and are thinking about taking lecanemab should be aware that there are hazards involved as well as only minor, often imperceptible benefits.